How many plots and what size of plots should I use for sampling plant communities?
A common plot size for sampling vegetation is 10 x 10 meters, which is large enough for sampling trees and understory plants, but not so large as to be difficult to sample or replicate. There is no magic size that is inherently better than another. You want to select a plot size that is large enough to capture some of the variability in your plant community (usually at least 1m2 for understory plants), but not so large that it becomes impractical. The size and distribution of the particular plants of interest also affect the plot size. For example, if you are sampling trees, you need a much larger plot than if you are only sampling small understory plants. Sometimes it is necessary to use one sized plot for some plants (like trees) and another size plot for others (like understory plants).
Perhaps more important than the size of the plots, is the total area sampled. You want to sample enough plots so that the total area sampled accurately captures the composition and diversity of the plant community of interest. Calculating a species-area curve for your study site is a very good way to assure that this goal is met (look below to read more about species-area curves).
Species Diversity
Plant diversity seems like a simple thing to measure, simply count the number of species, right? Well, yes and no. For example, say there are two sites that each contains 15 plant species. In one site, two of those species account for 90% of the total plant community with the other 13 species being present at very low levels, while in the other site, all 15 species are about equally abundant. Do these sites have the same diversity?
Sometimes, it is desirable to have a measure of diversity that includes both richness (number of species present) and their relative abundance. Several indices of diversity have been developed to help you do just that (see table 3 below). One that is very commonly used in plant communities is the Shannon Diversity Index, indicated by the symbol H’. I can be very interesting to see how the different measures of diversity can sometimes give different impressions of the same plant community!
| Index* | References | Emphasis |
| Richness | n/a | Number of Species |
| Shannon's H' =[-Σp)]-[(s-1)/2N] | Shannon and Weaver 1964; Poole 1974 | Integrates the number of species and relative abundance; derived from information theory; measure of entropy for the sample; full formula is and expanding series; the first two terms are shown here. |
| Relative H' = H'/ln(s) | Magurran 1988 | Represents the evenness of the relaive abundance of the species present; the percentage of relative entropy for a system. |
| Simpson's D= Σ² pi | Magurran 1988 | A measure of dominance by one or a few species; the probability that two individuals selected from the population at random will be of the same species. |
| Margalef: (s-1)/(ln N) | Magurran 1988 | Derived by fitting data; no theoretical explanation beyond how author believes species number and relative abundance may be related. |
| Menhinick: sl(N)^0.5 | Magurran 1988 | Derived by fitting data; no theoretical explanation beyond how author believes species number and relative abundance may be related. |
| Hulbert's: [Nl(N-1)]/(1-Σ pi² ) | Washington 1984 | Related to H' and D above; the probability of interspecific encounters |
*pi, proportion of N made up by the ith species; s, number of species recorded; N, total number of individuals. Table 3. Diversity Indices from ‘Hale et al 1999’
Plant Abundance
The two most common measures of plant abundance include stem counts and percent cover. Each has their pro’s and con’s. Depending on the questions you are asking, sometimes it is desirable to use both.
Stem Counts
Stem counts are accurate when determining the population size of a particular plant species, but can be time consuming in areas with dense plant communities. Plus it is always a challenge to figure out how to count clonal species like grasses that have many stems for each individual. Also, individuals of different species can vary a lot in size! For example, when using stem counts, a small violet is counted the same as a very large fern. When examining plant population dynamics within species, stem counts are the way to go. But when looking at overall plant communities, sometimes a measure of “how much space” a particular species occupies is a valuable thing to know. That’s where percent cover can be helpful.
Percent Cover
Percent cover is a method of determining relative abundance based on the amount of space they take up. In this method, rather than counting the number of individuals you assign each species to a “percent cover class” based on a visual estimate of how much of the sample plot they occupy (see figure 1 below). The reason most scientists use cover classes, rather than assigning a species number, like 16% or 48%, is that most people would not select the same number if they were looking at the same sample plot, it’s just too subjective. However, research has shown that most of the time, most people will select the same cover class when looking at the same sample plot, so it is a more reliable measure of plant abundance.
The Braun-Blanquet cover/abundance scale (adapted from “Aims and methods of vegetation ecology” by D. Mueller-Dombois and H. Ellenberg. 1974. Wiley New York) is perhaps the most commonly used in plant ecology:
|
Cover Class
|
Percent Cover
|
Description
|
|---|---|---|
| r | < 5% | Assigned to a species where there is only a single individual of the species (a plant with multiple stems arising from the same root would be classified as a single individual) and covers less than 5% of the sample plot area. |
| + | < 5% | Assigned to a species where there are only a few (approximately 2-20) individuals of the species and those individuals collectively cover less than 5% of the sample plot area |
| 1 | < 5% | Assigned to a species where there are numerous individuals of the species, but those individuals collectively cover less than 5% of the sample plot area |
| 2 | 5% - 25% | Assigned to a species where that species’ cover is between 5% and 25% of the sample plot area. |
| 3< | 25% - 50% | Assigned to a species where that species’ cover is between 25% and 50% of the sample plot area. |
| 4 | 50% - 75% | Assigned to a species where that species’ cover is between 50% and 75% of the sample plot area. |
| 5 | 75% - 100% | Assigned to a species where that species’ cover is between 75% and 100% of the sample plot area. |
Species Area Curves
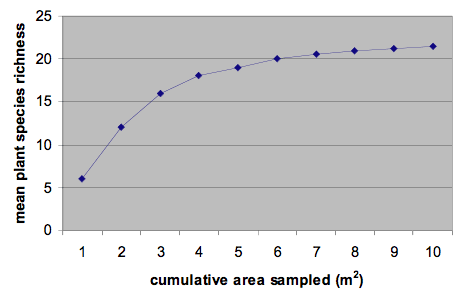
How do you know if you have collected enough samples of the plant community? You want to collect enough samples so that you don’t miss a lot of species, but since time and energy are limited you usually don’t want to sample too many more plots that necessary. Scientists use the species-area curve to answer the question of how many plots to sample.
| Number of Plots Sampled | Cumulative Area Sampled (m²) | Mean Plant Species Richness |
|---|---|---|
| 1 | 1 | 6 |
| 2 | 2 | 12 |
| 3 | 3 | 16 |
| 4 | 4 | 18 |
| 5 | 5 | 19 |
| 6 | 6 | 20 |
| 7 | 7 | 20.5 |
| 8 | 8 | 21 |
| 9 | 9 | 21.5 |
As the cumulative area of a study site sampled increases, the number of species detected increases and then begins to plateau (see Figure 1 below). Once that plateau is reached, you can generally be confident that an appropriate area has been sampled in order to adequately capture the majority of species present in a community. A species-area curve plots the average number of species detected against the cumulative area sampled. For example, table 1 lists data from a study that used 1.0 m2 plots where all plant species present in each plot were identified. As the cumulative area sampled increases the mean (average) plant species richness also increases. In this case plot 1 contained 6 species; plot 2 contained 18 species, so the mean (average) number of species was 12, and so on. You can see in figure 1 below, that the rate of increase in the number of species begins to plateau after 6 plots are sampled and is pretty stable once 10 plots are sampled, indicating that this is probably enough samples to accurately represent the composition and diversity of this plant community. Different habitats and plant communities will have different species area curves, so just because 10 plots were enough in this site doesn’t mean this is true for all sites. Some sites may require fewer plots, others more.